Soil pH is the measure of acidity and alkalinity in soil chemistry. Plant nutrients are affected by soil pH as the pH controls the nutrient’s chemical form.
Knowing the texture of your soil helps you understand more about how the soil holds moisture and encourages root growth and development.
Acidity and pH
Soils can be acid or alkaline. Soils in the higher rainfall areas (above 600 mm per year) would rather develop acid characteristics, while alkaline soils occur largely in the lower rainfall areas. This would be the common tendency, but it is not always the case as more factors, other than rainfall, might determine soil pH. The soil pH expresses the degree of soil acidity on a scale from 1 (highest acidity) through 7 (neutrality) to 14 (highest alkalinity) as indicated in Figure 1.
Soil pH is of utmost importance in plant growth as it influences nutrient availability, toxicities, and the activity of soil organisms. Acidification of soils results in a gradual decline in yields. Some plants are tolerant to acidic soils, for example, rice and tea, but most of them grow better in neutral or slightly alkaline soils. The level of acidity that plants can tolerate is influenced by the supply of available nutrients and moisture.
If the pH is too low, in other words, the soil is too acidic, lime could be applied under irrigation circumstances. With dry-land production, this investment is not worth it, and one should rather find a crop that tolerates the low pH. If the pH is too high, meaning the soil is too alkaline, acidifying fertilisers such as ammonium sulphate, ammonium nitrate, MAP, or acidifying improvers such as compost made of pine needles should be applied.
Figure 1: The pH scale.
Salinity and alkalinity – sodicity
Soluble salts are found in all soils and natural waters. However, sometimes these salts accumulate in soils to such an extent that they can impair proper plant growth. Salts accumulate in soils largely because of infiltration of the runoff of irrigation water, together with evapotranspiration, which concentrates these waters. The salts are mainly chlorides bicarbonates, sulphates of sodium, potassium, calcium and magnesium.
The salt concentration of soils is measured by electric conductivity (EC) and the unit with which the EC is expressed in soil analysis results is mS/cm (millisiemens per centimetre).
Alkaline (sodic) soils are associated with high sodium content and a high pH in the absence of surplus soluble salts. The EC is generally less than 4 mS/cm, and the pH is 8,5 or more.
Figure 2: Soil texture.
Plant growth is negatively affected by sodic conditions because of:
- high pH that leads to nutritional imbalances;
- toxicity of specific ions such as carbonates, sodium and molyb-denum; and
- poor drainage and aeration because of the soil forming a crust.
Reclamation of sodic soils essentially requires extensive leaching of the sodium salts in the soil and replacing these with calcium salts like gypsum or acidifying agents like sulphuric acid or iron sulphate. Manure or compost will improve the soil structure and therefore the tolerance of crops to sodic soils will be improved due to better aeration as well.
Under dry-land conditions, it is advisable to opt for a crop that can tolerate the given soil conditions rather than try to change the soil. Planting an unsuitable crop will result in more losses than not planting at all!
Soil texture
Soil particles are initially divided into two sizes: those larger than 2 mm in diameter, usually called gravel, and those smaller than 2 mm in diameter, called fine earth. Soil texture can be defined as the relative proportions of sand, silt and clay within the fine earth. Sand particles can be seen individually with the naked eye. Sandy soils are well aerated and have good drainage but will not hold water or nutrients very well.
Silt particles can be seen by using a microscope. Silt contains less silica than sand and has better nutrient-holding characteristics than sand.
Clay particles can only be seen by using a microscope. Clay particles (or colloids) are important because they can hold nutrients on their surface in a form that is readily available to plants. They can also hold water better than sand. On the negative side, clay soils tend to become waterlogged in the rainy season, causing poor aeration and subsequently, poor root development. Clay is sticky when wet and may form very hard clods when dry.
Sand, silt and clay percentages are obtained by carrying out a particle size analysis of a sample in the laboratory.
Farmers often describe clay soils as “heavy” and sandy soils as “light”. This does not refer to their weight, but to their ease of working. A heavy soil requires much more effort to cultivate than a light soil. Medium textured soils (such as loam, clay loam, sandy clay loam, silty clay loam, sandy loam and silt loam) are best for most plants. Organic matter (compost and kraal manure) should be incorporated into the soil to improve the soil texture.

Testing soil composition with a pH testing kit.
Soil fertility evaluation
Soil sampling
The analysis is no better than the sample. This means that sampling must be done very carefully to ensure that the sample taken is typical of what it is meant to represent. Nowadays, laboratory procedures for soil analysis are relatively standardised and accurate, and the main source of error often comes from the initial sampling of the soil. No effort or careful laboratory work is of any use at all if the sample is not properly taken. The area to be sampled must be uniform as regards slope, drainage, soil colour and texture, and history of cropping and fertiliser use. If a field includes obviously different areas, it must be subdivided for sampling purposes. The soil is sampled from at least 10 to 20 sites per subdivision.
All these samples are then thoroughly mixed to form one big composite sample. Out of this big sample, one representative sample of about 1 kg is taken and sent to the laboratory.
For sampling purposes, the soil is generally subdivided into the topsoil (0 – 30 cm deep) and the subsoil (30 – 60 cm deep). For deep-rooted crops, a representative subsoil (which is once again a composite of 10 to 20 subsoil samples) should be taken and sent as such to the laboratory as well.
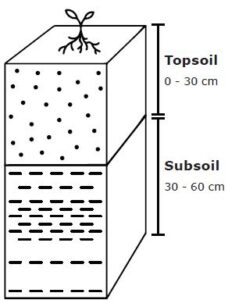
Figure 3: Soil sampling.
Soil analysis interpretation and fertiliser application
Once a soil sample has been analysed by the agricultural laboratory in Windhoek, soil analysis results will be returned to the applicant. Depending on the request by the applicant, these will include the respective concentrations of individual nutrients in the soil as well as pH and soil texture percentages of sand, silt and clay. Only the features as indicated on the application form will be analysed.
As soon as the soil analysis results are available, fertiliser recommendations can be made. This is a highly specialised task, which requires a lot of experience and knowledge. In practice, most farmers would be expected to consult experts before expensive fertilisers are purchased and applied in noticeable quantities.
The information in this article is credited to the Namibia Agricultural Union and Namibia National Farmers Union who published the Crop Production Manual in 2008.









